|
The chemical formula of ammonia is NH 3 3 3 start subscript, 3, end subscript. Sn 2+ (aq) +2e - → Sn (s) with SRP E o = -0.137 V (Cathode where reduction happens)Ģ. Metal sequestering by different parts of the cell can occur via various processes: complexation, chelation, coordination, ion exchange, precipitation. Ionic bonding occurs between a metal and a non-metal The metal has a nearly empty outer shell and so loses electrons to form a positively charged cation The. Oxygen is a non-metal element that is a gas at room temperature. They primarily form coordinate covalent bonds, a form of the Lewis acid-base interaction in which both. Ionic Bond (Electrovalent Bond) - Ionic bonding involves the electrostatic interaction between oppositely charged species. The behavior of these ions produces the tracings of heart and brain function observed as waves on an electrocardiogram (EKG or ECG) or an electroencephalogram (EEG). The crystals have ionic bonding, and each ion has six or eight neighbours. These dissolved ions produce electrical charges within the body. Another important observation is that, in the absence of metal ions. To determine oxidation electrodes, the reduction equation can simply be flipped and its potential changed from positive to negative (and vice versa).\) for the voltaic cell formed by each reaction.ġ.a) Ba 2+ (aq) → Ba (s) + 2e- with SRP (for opposite reaction) E o = -2.92 V (Anode where oxidation happens)Ĭu 2+ (aq) + 2e- → Cu (s) with SRP E o = +0.340 V (Cathode where reduction happens)ġ.b) Al 3+ (aq) → Al (s) + 3e - with SRP (for opposite reaction) E o = -1.66 V (Anode where oxidation happens) Transition metals do not normally bond in this fashion. In fact, in biological fluids, most individual atoms exist as ions. Thus interchain hydrogen bonds destabilize metal ion binding by 812 kcal/mol (see below). 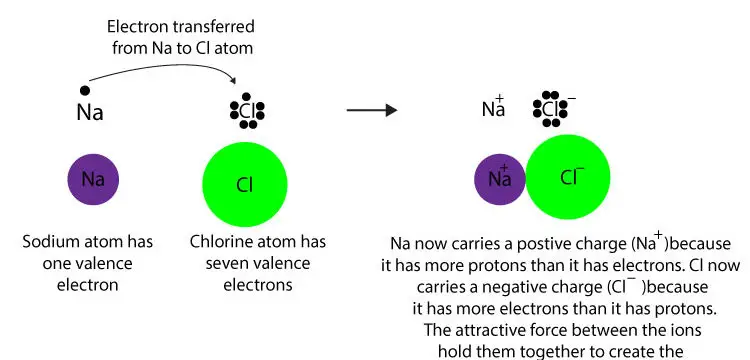
The bond between the metal ion and the ligand, where the ligand supplies both electrons, is known as a coordinate covalent bondSimple ligands include water, ammonia and chloride ions. The coordination numberis the number of places on the metal ion where ligands are bound. 
The table below is a list of important standard electrode potentials in the reduction state. The anions or molecules attached to the metal are called ligands. What is metallic bonding in A-level Chemistry In A-level Chemistry, metallic bonding refers to the chemical bonding that occurs between metal atoms in a. Important Standard Electrode (Reduction) Potentials The attraction between oppositely charged ions is called an ionic bond, and it is one of the main types of chemical bonds in chemistry. We could have accomplished the same thing by taking the difference of the reduction potentials, where the absent or doubled negation accounts for the fact that the reverse of the reduction reaction is what actually occurs.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
